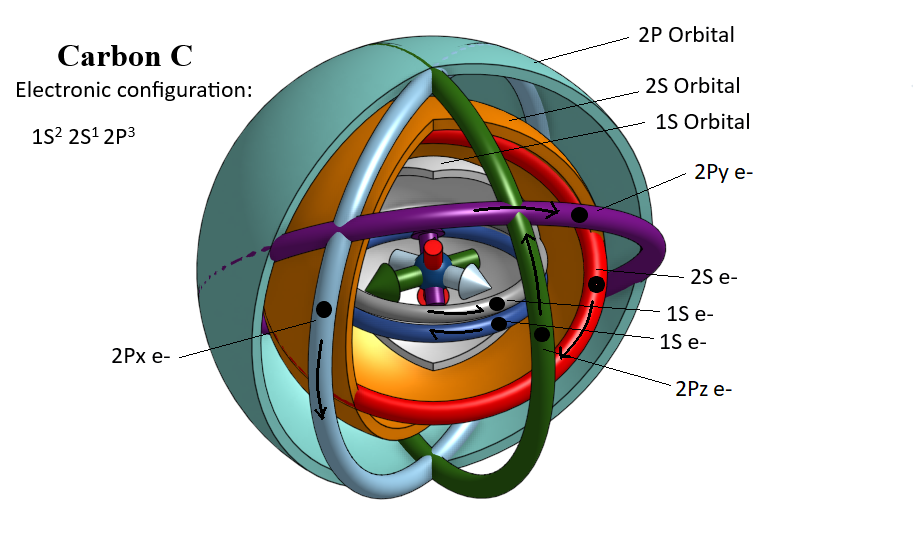
THE METALLIC BOND IS A MAGNETIC BOND.
Εxplanation of the metallic bond with Neodymium (Nd) spherical magnets.
In the present study we try to explain the metallic bond with the use of spherical Neodymium magnets.
Maybe at first it sounds a little crazy, but the information I'm going to quote later will surprise you.
The magnetic behavior of Neodymium spherical magnets can produce all the bonding patterns observed in metal surface images by scanning electron microscopes, as well as three dimensional (3D) metallic bond models (SP, BCC, CCP, HCP, Silicon, Grafene, Grafite, Carbon Nanotybes, Fullerences, Quasicrystals ......).
In the present study we try to explain the metallic bond with the use of spherical Neodymium magnets.
Maybe at first it sounds a little crazy, but the information I'm going to quote later will surprise you.
The magnetic behavior of Neodymium spherical magnets can produce all the bonding patterns observed in metal surface images by scanning electron microscopes, as well as three dimensional (3D) metallic bond models (SP, BCC, CCP, HCP, Silicon, Grafene, Grafite, Carbon Nanotybes, Fullerences, Quasicrystals ......).
|
|
|
|
Read more... GRAPHENE
|
Read more... CARBON NANOTUBES
|
|
|
|
|
Read more... CARBON NANOTUBES
|
Read more... FULLERENES
|
|
|
|
|
Read more... SILICON
|
Read more... BBS BIBI SPRING
|
|
Pentagonal bipyramid
|
Elongated pentagonal bipyramid
|
George Georgitzikis
Professor of Geology
Mytilini Lesvos Greece.
15 June 2017
All rights reserved
Professor of Geology
Mytilini Lesvos Greece.
15 June 2017
All rights reserved
An experiment that strengthens the theory of magnetic bonding.
How to melt gold at room temperature.(21-11-2018)
As tension rises unexpected things can happen. This is also true for gold atoms. Researchers, have, for the first time, managed to make the surface of a gold object melt at room temperature.
Watch the entire video:
www.facebook.com/jyunanoscience/videos/1904246952958010/?t=5
How to melt gold at room temperature.(21-11-2018)
As tension rises unexpected things can happen. This is also true for gold atoms. Researchers, have, for the first time, managed to make the surface of a gold object melt at room temperature.
Watch the entire video:
www.facebook.com/jyunanoscience/videos/1904246952958010/?t=5
Ludvig de Knoop, from Chalmers’ Department of Physics, placed a small piece of gold in an electron microscope. Observing it at the highest level of magnification and step-by-step increasing the electric field to extremely high levels he was to study how it influenced the gold atoms. When studying the atoms in the recordings from the microscope, he saw something exciting. The surface layers of gold atoms had actually melted, at room temperature.
- I was really stunned by the discovery. This is an extraordinary phenomenon, and it gives us new, foundational knowledge of gold, says Ludvig de Knoop.
What happened was that the gold atoms became excited. Under the influence of the electric field, they suddenly lost their ordered structure and released almost all their connections to each other. Upon further experimentation, the researchers also discovered that it was possible to switch between a solid and a molten structure.
The discovery of how gold atoms can lose their structure in this way is not just spectacular, but also groundbreaking scientifically. Ludvig de Knoop, together with the theoretician Mikael Juhani Kuisma and coworkers, have opened up new avenues in materials science. The results are now published in the journal Physical Review Materials. Thanks to theoretical calculations, the researchers are able to suggest why gold can melt at room temperature. Possibly, the surface melting can be seen as a so-called low-dimensional phase transition. In that case, the discovery is connected to the research field of topology, where pioneers David Thouless, Duncan Haldane and Michael Kosterlitz received the Nobel Prize in Physics 2016. With Mikael Juhani Kuisma in the lead, the researchers are now looking into that possibility. In any case, the ability to melt surface layers of gold in this manner enables various novel practical applications in the future.
- Because we can control and change the properties of the surface atom layers, it opens doors for different kinds of applications. For example, the technology could be used in different types of sensors, catalysts and transistors. There could also be opportunities for new concepts for contactless components, says Eva Olsson, Professor at the Department of Physics at Chalmers.
For now, those who want to melt gold without an electron microscope, a trip to the goldsmith is still a necessity.
Text: Joshua Worth ([email protected]) and Mia Halleröd Palmgren ([email protected])
For more information:
ContactsUniversity Marketing and Communications:
[email protected]
+358 40 805 4403
- I was really stunned by the discovery. This is an extraordinary phenomenon, and it gives us new, foundational knowledge of gold, says Ludvig de Knoop.
What happened was that the gold atoms became excited. Under the influence of the electric field, they suddenly lost their ordered structure and released almost all their connections to each other. Upon further experimentation, the researchers also discovered that it was possible to switch between a solid and a molten structure.
The discovery of how gold atoms can lose their structure in this way is not just spectacular, but also groundbreaking scientifically. Ludvig de Knoop, together with the theoretician Mikael Juhani Kuisma and coworkers, have opened up new avenues in materials science. The results are now published in the journal Physical Review Materials. Thanks to theoretical calculations, the researchers are able to suggest why gold can melt at room temperature. Possibly, the surface melting can be seen as a so-called low-dimensional phase transition. In that case, the discovery is connected to the research field of topology, where pioneers David Thouless, Duncan Haldane and Michael Kosterlitz received the Nobel Prize in Physics 2016. With Mikael Juhani Kuisma in the lead, the researchers are now looking into that possibility. In any case, the ability to melt surface layers of gold in this manner enables various novel practical applications in the future.
- Because we can control and change the properties of the surface atom layers, it opens doors for different kinds of applications. For example, the technology could be used in different types of sensors, catalysts and transistors. There could also be opportunities for new concepts for contactless components, says Eva Olsson, Professor at the Department of Physics at Chalmers.
For now, those who want to melt gold without an electron microscope, a trip to the goldsmith is still a necessity.
Text: Joshua Worth ([email protected]) and Mia Halleröd Palmgren ([email protected])
For more information:
- Postdoctoral Researcher Mikael Kuisma (University of Jyväskylä), [email protected]
- Postdoctoral researcher Ludvig de Knoop (Chalmers University of Technology, Sweden), +46 31 772 51 80, [email protected]
ContactsUniversity Marketing and Communications:
[email protected]
+358 40 805 4403
The author's opinion:
I think the explanation is very simple. As in the Oersted experiment, when applying an electric field near a magnetic compass it deviates from its position, so in this case the application of a strong electric field disorients the magnetic field of the atoms of the gold and forces them into sublimation. That is, the magnetic force that joins them is lost and the atoms spreads to the surrounding space.
George Georgitzikis
19-4-2019
I think the explanation is very simple. As in the Oersted experiment, when applying an electric field near a magnetic compass it deviates from its position, so in this case the application of a strong electric field disorients the magnetic field of the atoms of the gold and forces them into sublimation. That is, the magnetic force that joins them is lost and the atoms spreads to the surrounding space.
George Georgitzikis
19-4-2019
SEE ALSO A NEW THEORY !!!!!
We sincerely thank.
The creation and processing of the 3D models was done with the program Autodesk Inventor Professional 2016, after the courtesy licensing of the company Autodesk.
The video was created and edited with the program FlashBack Express Recorder, after the courtesy licensing of the company Blueberry Software.
We also greatly thank companies Weebly and YouTube for free web hosting.
The creation and processing of the 3D models was done with the program Autodesk Inventor Professional 2016, after the courtesy licensing of the company Autodesk.
The video was created and edited with the program FlashBack Express Recorder, after the courtesy licensing of the company Blueberry Software.
We also greatly thank companies Weebly and YouTube for free web hosting.
|
|
|