|
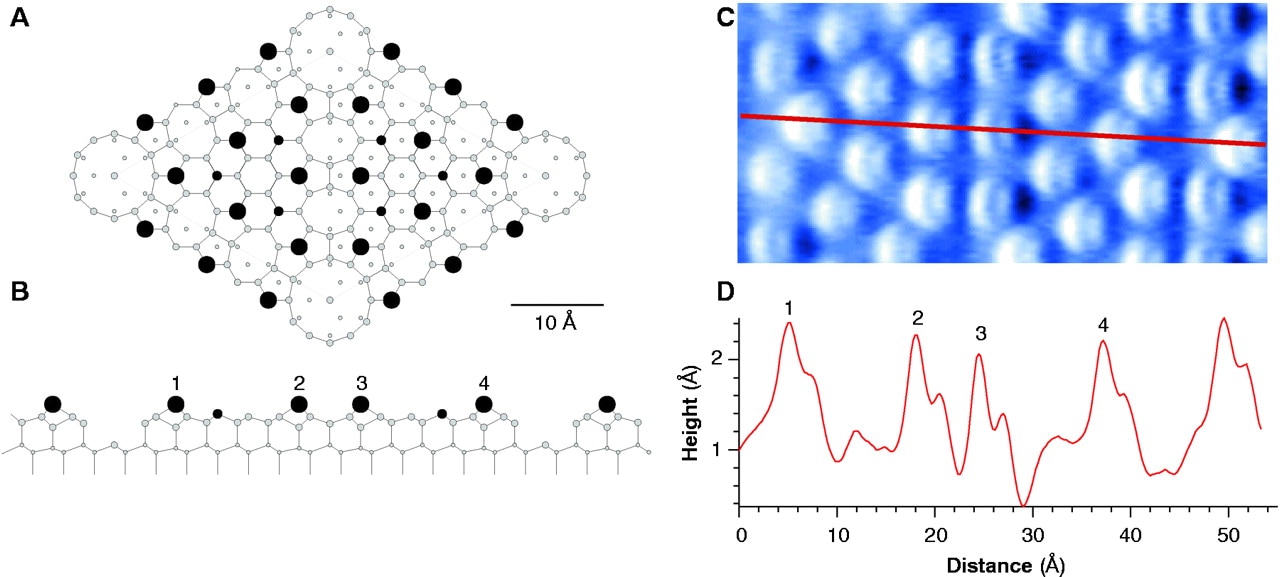
Silicon is a metalloid element and its atoms appear to be magnetically bonded. Everything starts with the hexagonal ring of six silicon atoms. If lateral forces are applied to the ring at 120 degrees, the hexagonal ring will be converted into a triangle of six silicon atoms. These triangles interconnected together give the structure of the silicon seen in the electron microscope images. Exagon ring of six silicon atoms. Lateral forces are applied to the ring at 120 degrees. Triangle of six silicon atoms. Silicon structure. Silicon structure and six triangles. Observe below the resemblance to electron microscope images. Subatomic Features on the Silicon (111)-(7×7) Surface Observed by Atomic Force Microscopy Franz J. Giessibl, S. Hembacher, H. Bielefeldt and J. Mannhart (July 21,2000) http://science.sciencemag.org/content/289/5478/422 Subatomic Features on the Silicon (111)-(7×7) Surface Observed by Atomic Force Microscopy Franz J. Giessibl, S. Hembacher, H. Bielefeldt and J. Mannhart (July 21,2000) http://science.sciencemag.org/content/289/5478/422 This video contains English subtitles.
0 Comments
|



 RSS Feed
RSS Feed
