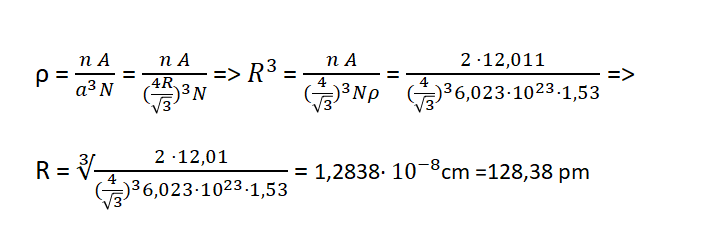THE MAGNETIC BOND THEORY
The unification of all chemical bonds.
Magnetic bond theory is a theory that attempts to explain all chemical bonds with magnetic force. It is at the same time an attempt to unify all chemical bonds.
The pre-existing theories of valence bonds as well as the quantum theory of atomic and molecular orbits are two wonderful theories but they present some vague points and some weaknesses.
With the new theory of magnetic bonding, it is not necessary to distinguish between metal and non-metallic or metalloids, as well as the concepts of electronegativity and electropositivity of the elements, but also the positive and negative valence numbers.
With the new theory of magnetic bond, I will try to illuminate these vague points and I think we will take a step closer to the truth.
I accept the existence of atomic and molecular orbits, as well as the links that develop between them. My disagreement lies in the overlapping of atomic and molecular orbits and the kind of bond created between them.
The basic differentiation of the magnetic bond theory lies in the fact that when two atomic or molecular orbits approach each other, then there is no overlap of the orbits, but a repulsion of the electron cloud so that it accumulates in a central zone of each atom and therefore, the emergence of a magnetic field with north and south pole.
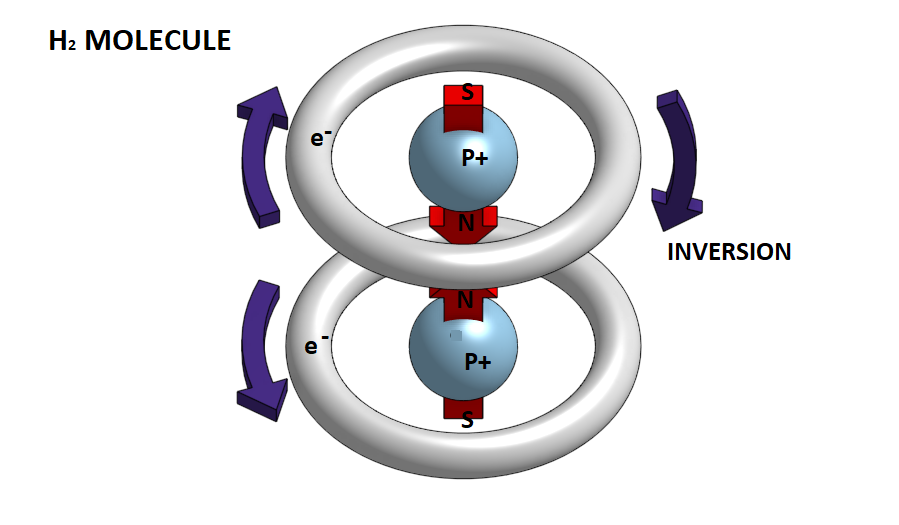
Let's look at the simplest case of the hydrogen molecule Η2.
The pre-existing theories of valence bonds as well as the quantum theory of atomic and molecular orbits are two wonderful theories but they present some vague points and some weaknesses.
With the new theory of magnetic bonding, it is not necessary to distinguish between metal and non-metallic or metalloids, as well as the concepts of electronegativity and electropositivity of the elements, but also the positive and negative valence numbers.
With the new theory of magnetic bond, I will try to illuminate these vague points and I think we will take a step closer to the truth.
I accept the existence of atomic and molecular orbits, as well as the links that develop between them. My disagreement lies in the overlapping of atomic and molecular orbits and the kind of bond created between them.
The basic differentiation of the magnetic bond theory lies in the fact that when two atomic or molecular orbits approach each other, then there is no overlap of the orbits, but a repulsion of the electron cloud so that it accumulates in a central zone of each atom and therefore, the emergence of a magnetic field with north and south pole.
Let's look at the simplest case of the hydrogen molecule Η2.
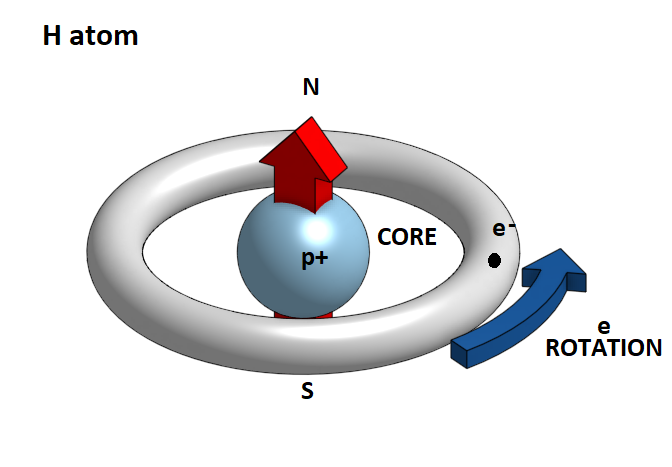
In this case the unique electron generates 1s orbital. The distribution of the electronic cloud resembles a sphere in which the unique electron moves.
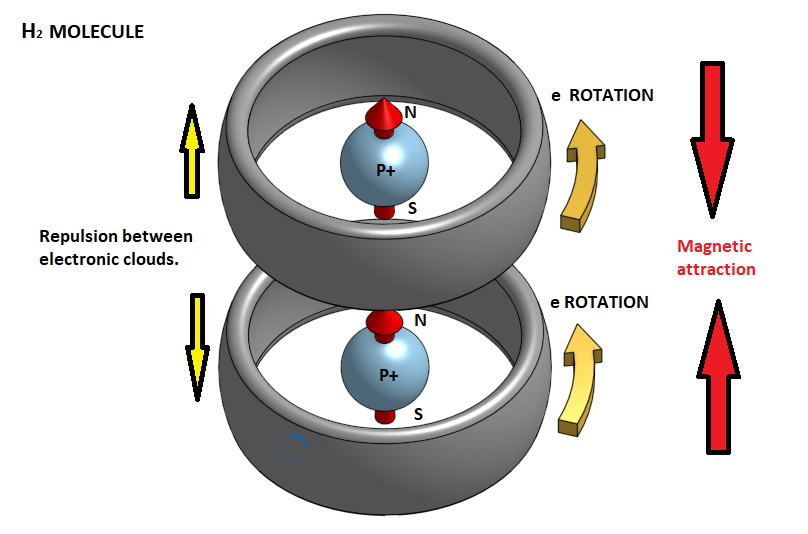
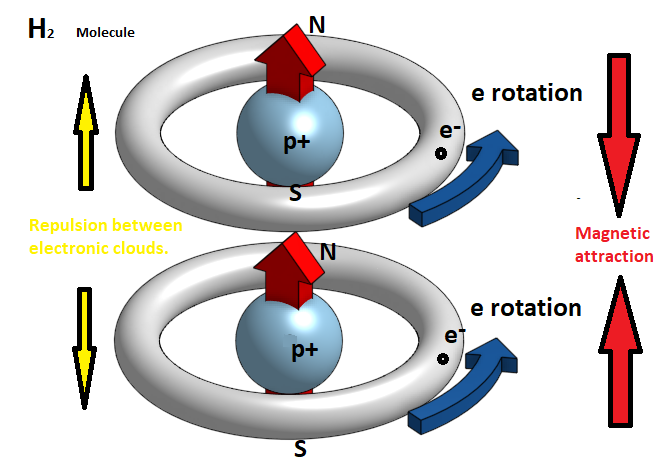
In fact, the electron clears circular trajectories around the atom's nucleus and therefore at any moment creates a magnetic field with a particular polarity. However, since we have a continual alternation of this rotational motion so that at each moment the electron is in a different orbit, the atom as a whole behaves like a neutral magnetic body. So when two hydrogen atoms approach each other the electronic clouds of 1s orbits that resemble two negatively charged spheres, they are repelled. But if atoms are forced to move closer to one another because of the increase in pressure in a gas or a conflict between them, then the electronic clouds of the two atoms are repelled and approaching a central point of the atom, creating two distinct bands of rotation. At the same time, an oriented magnetic field appears, which now prevails over the electrostatic forces, and pulls the two atoms so as to form a cross-sectional molecule. This is the hydrogen molecule H2.
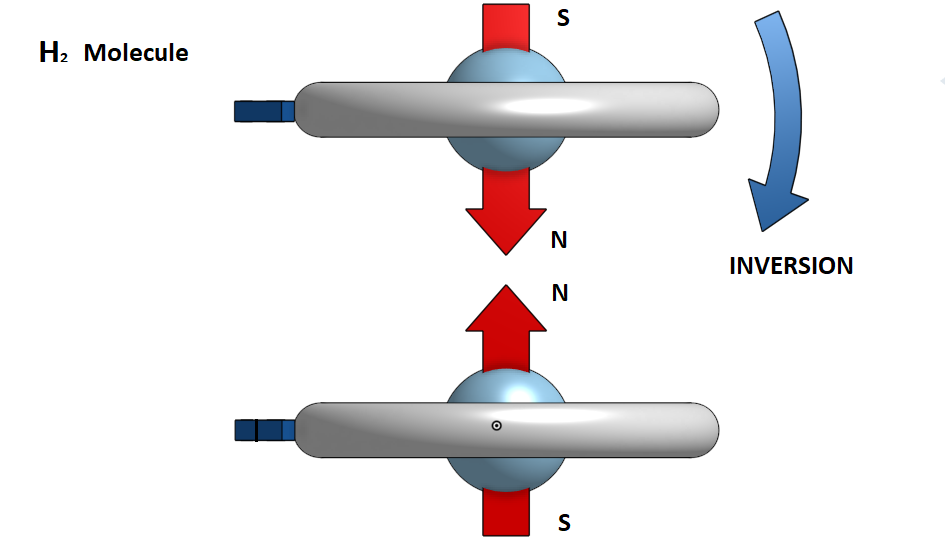
In the hydrogen molecule H2 the two electrons are homogeneous. In case when the two atoms approach the electrons have opposite turns, then one of the two is reversed so that they become homogeneous and have a magnetic attraction between the north and south magnetic poles.
Obviously, to do this, pressure conditions should be very high and the temperature too low. In this case, when such magnetic dipoles of hydrogen molecules are created, it is possible to join them together and form a metal bond. That is, we will have the metallic hydrogen.
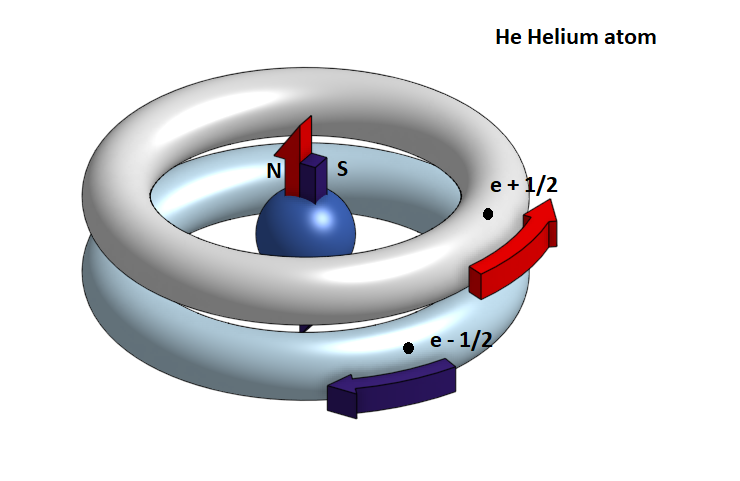
INTERPRETATION OF THE STRUCTURE OF THE HELIUM ATOM He.
In the Helium atom He, there are two protons and two neutrons in the nucleus and two electrons that roam in the K layer. These electrons have an opposite spin, that is, they rotate in opposite directions in parallel circular paths, creating opposite magnetic fields attracted to each other and mutually eliminated. Thus the orbital S generated does not display a magnetic field and remains magnetically neutral.
In the Helium atom He, there are two protons and two neutrons in the nucleus and two electrons that roam in the K layer. These electrons have an opposite spin, that is, they rotate in opposite directions in parallel circular paths, creating opposite magnetic fields attracted to each other and mutually eliminated. Thus the orbital S generated does not display a magnetic field and remains magnetically neutral.
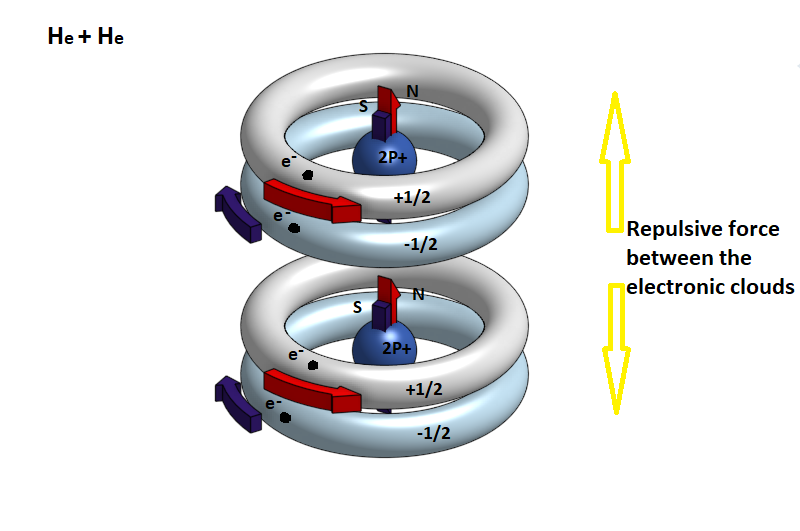
It is therefore very difficult, when two helium atoms approach, to join the atoms to the molecule, because the two electrons of the S orbits are strongly repulsed by the negative electron load, while there is no magnetic attraction.
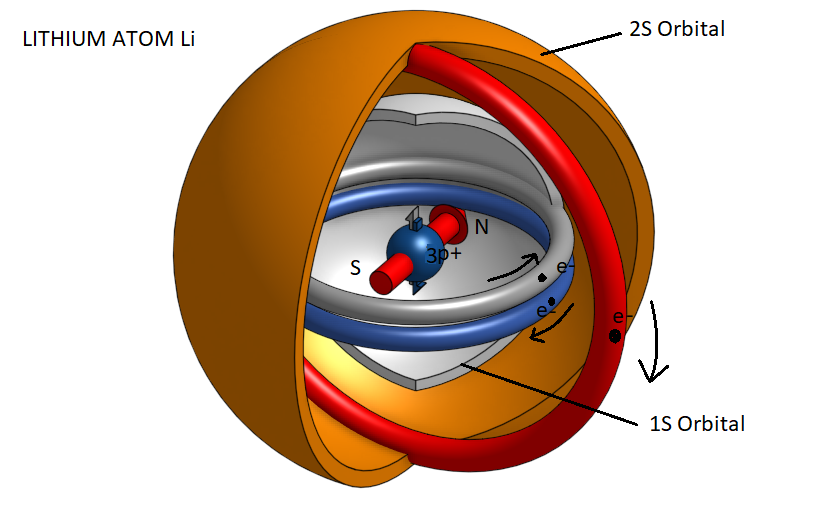
Interpretation of the structure of the lithium atom Li .
Lithium is the third element of the periodic table and has the following electronic configuration 1s2 2s1. That is, it has a completed layer K with two electrons and a single electron in the L layer.
As we observe because in the K layer there are two electrons in the opposite direction of rotation, their magnetic fields are mutually exclusive. However, in the outer layer L where an electron exists, a pure magnetic dipole is created due to the rotation of the electron. Thus the lithium atom appears as a magnetic dipole and is therefore capable of forming strong bonds with other lithium atoms such as the metallic bond in the lithium metal. This magnetic bipolarity also explains its active character, that is, the ability of lithium to form strong bonds with other atoms in the periodic table.
Interpretation of the structure of the Beryllium atom Be.
Beryllium is the fourth element of the periodic table and has the following 1S2 2S1 2P1 electronic configuration. That is, it has a completed layer K with two electrons and two single electrons in the L layer, one in the 2S sublayer and the other in the 2Px sublayer. In the literature, the following 1S2 2S2 electronic configuration is mentioned, which however leads to a noble gas structure and obviously Be should be an inactive element, which is not the case. The magnetic dipoles formed by the 2S1 and 2P1 electron sweep around the core are repelled together to align with opposite polarity. This would, of course, lead to the neutralization of the magnetic momentum of the two dipoles. However, because the magnetic moment of the electron 2S1 (red arrow) is less than the magnetic moment of the electron 2P1 (light blue arrow), the overall magnetic moment is the difference between them, in the opposite direction to that of the 2P1 electron. Therefore, Be exhibits bipolar momentum and can form bivalent compounds with aligned bonds as in the case of BeCl2. But these bonds are magnetic. The electron pair 1S2 shows no magnetic moment due to neutralization.
Beryllium is the fourth element of the periodic table and has the following 1S2 2S1 2P1 electronic configuration. That is, it has a completed layer K with two electrons and two single electrons in the L layer, one in the 2S sublayer and the other in the 2Px sublayer. In the literature, the following 1S2 2S2 electronic configuration is mentioned, which however leads to a noble gas structure and obviously Be should be an inactive element, which is not the case. The magnetic dipoles formed by the 2S1 and 2P1 electron sweep around the core are repelled together to align with opposite polarity. This would, of course, lead to the neutralization of the magnetic momentum of the two dipoles. However, because the magnetic moment of the electron 2S1 (red arrow) is less than the magnetic moment of the electron 2P1 (light blue arrow), the overall magnetic moment is the difference between them, in the opposite direction to that of the 2P1 electron. Therefore, Be exhibits bipolar momentum and can form bivalent compounds with aligned bonds as in the case of BeCl2. But these bonds are magnetic. The electron pair 1S2 shows no magnetic moment due to neutralization.
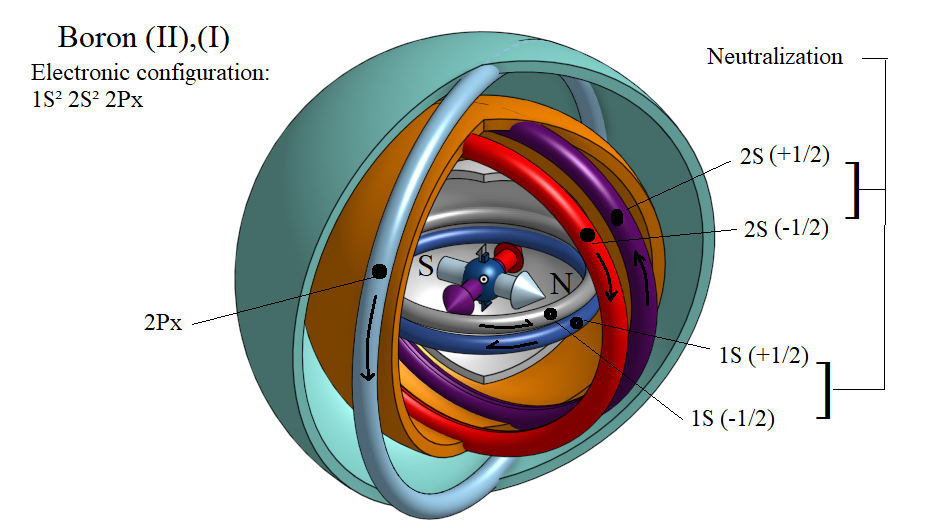
Interpretation of the structure of the Boron atom B.
Boron is the fifth element of the periodic table and has the following electronic configuration 1S2 2S1 2P2. That is, it has a completed K layer with two electrons and three single electrons in the L layer, one in the 2S sublayer, and the other two in the 2Px and 2Py sublayer. The following electronic configuration is mentioned in the literature: 1S2 2S2 2P1, but it cannot interpret the trivalent (± 3) character of the Βoron. The magnetic dipoles formed by the 2S1, 2P1x and 2P1y electrons around the core are repelled together to form angles of 120 ° in a planar configuration. The same would happen if I had three concentric rod-shaped magnets. The electron pair 1S2 shows no magnetic moment due to neutralization.
Boron is the fifth element of the periodic table and has the following electronic configuration 1S2 2S1 2P2. That is, it has a completed K layer with two electrons and three single electrons in the L layer, one in the 2S sublayer, and the other two in the 2Px and 2Py sublayer. The following electronic configuration is mentioned in the literature: 1S2 2S2 2P1, but it cannot interpret the trivalent (± 3) character of the Βoron. The magnetic dipoles formed by the 2S1, 2P1x and 2P1y electrons around the core are repelled together to form angles of 120 ° in a planar configuration. The same would happen if I had three concentric rod-shaped magnets. The electron pair 1S2 shows no magnetic moment due to neutralization.
When we have a 2Py electron transition in the 2S sublayer, then we have the following electronic configuration 1S2 2S2 2P1 , which explains the monovalent (± 1) or bivalent (± 2) character of the Βoron, because the only electron that generates magnetic moment is 2P1x. That is, there is an active magnetic dipole. The electron pairs 1S2 and 2S2 show no magnetic moment due to neutralization.
Interpretation of the structure of the Carbon atom C.
Carbon is the sixth element of the periodic table and has the following 1S2 2S1 2P3 electronic configuration. That is, it has a completed layer K with two electrons and four single electrons in the L layer, one in the 2S sublayer and the other three in the 2Px, 2Py and 2Pz sublayers. In the literature, the following 1S2 2S2 2P2 electronic configuration is mentioned but it cannot interpret the tetravalent (± 4) character of the Carbon. The four magnetic dipoles formed by the electron sweep 2S1, 2P1x, 2P1y and 2P1z around the core, are repelled together to form angles of 109.5 ° in 3D. The same would happen if I had four concentric rod-shaped magnets. The electron pair 1S2 shows no magnetic moment due to neutralization.
Carbon is the sixth element of the periodic table and has the following 1S2 2S1 2P3 electronic configuration. That is, it has a completed layer K with two electrons and four single electrons in the L layer, one in the 2S sublayer and the other three in the 2Px, 2Py and 2Pz sublayers. In the literature, the following 1S2 2S2 2P2 electronic configuration is mentioned but it cannot interpret the tetravalent (± 4) character of the Carbon. The four magnetic dipoles formed by the electron sweep 2S1, 2P1x, 2P1y and 2P1z around the core, are repelled together to form angles of 109.5 ° in 3D. The same would happen if I had four concentric rod-shaped magnets. The electron pair 1S2 shows no magnetic moment due to neutralization.
CONNECTION OF CARBON ATOMS IN THE DIAMOND
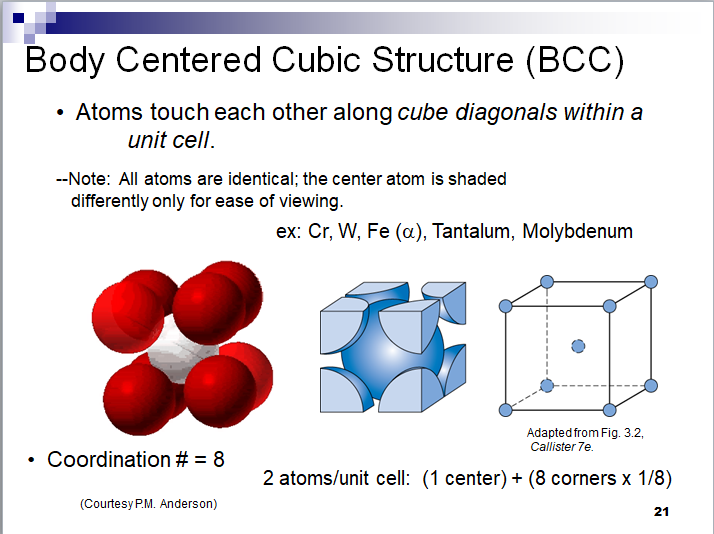
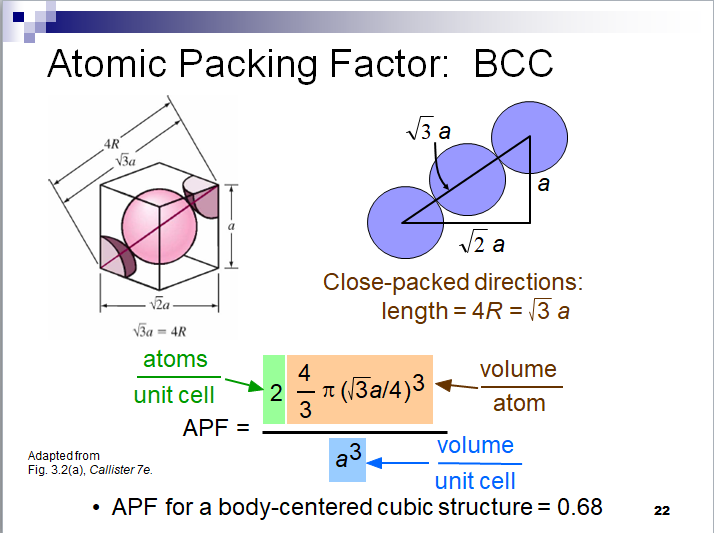
This is a new proposed structure for carbon in diamonds. Each carbon atom is linked to eight other carbon atoms by a strong magnetic bond. The structure that is created is of the type BCC with an APF of 68%. This structure explains the high hardness of the diamond and its transparency due to the gaps between the carbon atoms.
This is a new proposed structure for carbon in diamonds. Each carbon atom is linked to eight other carbon atoms by a strong magnetic bond. The structure that is created is of the type BCC with an APF of 68%. This structure explains the high hardness of the diamond and its transparency due to the gaps between the carbon atoms.
According to the existing theory in the BCC structure applies:
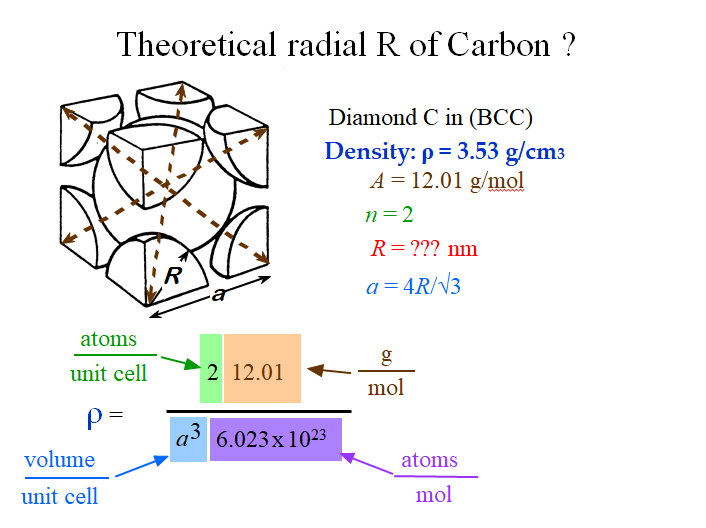
Calculation of the radius of the carbon atom in the diamond.
If diamond's density is ρ=1,53 gr/cm^3 then:
So we conclude that the atomic radius of carbon atoms in the diamond is R= 128,38 pm
The above suggestions are thoughts of the author that are in progress and are likely to be completed or modified in the future.
All rights reserved.
Mytilini Greece 18-5-2019
George Georgitzikis
All rights reserved.
Mytilini Greece 18-5-2019
George Georgitzikis