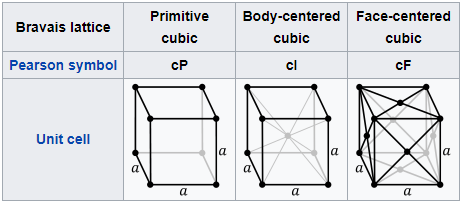
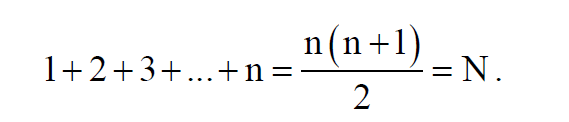CUBIC SYSTEM
REGULAR HEXAHEDRON
Cube in Simple Cubic.
Neodymium magnets can create cubic crystals in the Simple Cubic (SC) configuration.
Consists of successive layers of Simple Packing (SP) layout.
The Atomic Packing Factor is: APF=0,5236
The only metal crystallized in this structure is the Polonium (Po).
More information about this polyhedron can be found at the link below:
en.wikipedia.org/wiki/Cube
Consists of successive layers of Simple Packing (SP) layout.
The Atomic Packing Factor is: APF=0,5236
The only metal crystallized in this structure is the Polonium (Po).
More information about this polyhedron can be found at the link below:
en.wikipedia.org/wiki/Cube
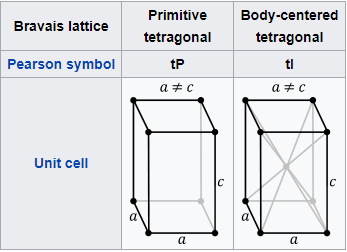
TETRAGONAL SYSTEM
TETRAHEDRON
It is a very compact structure consisting of equilateral triangular planes with a closed magnet arrangement.
Each triangular plane has a Closed Packing (CP) structure.
The cordination number is 12.
The unit cell expressing this type of crystallization is the Tetragonal (I) Body-Centered unit cell.
The crystallisation structure is CCP and the APF=0,74.
More information about this polyhedron can be found at the link below:
en.wikipedia.org/wiki/Tetrahedron
Each triangular plane has a Closed Packing (CP) structure.
The cordination number is 12.
The unit cell expressing this type of crystallization is the Tetragonal (I) Body-Centered unit cell.
The crystallisation structure is CCP and the APF=0,74.
More information about this polyhedron can be found at the link below:
en.wikipedia.org/wiki/Tetrahedron
|
|
|
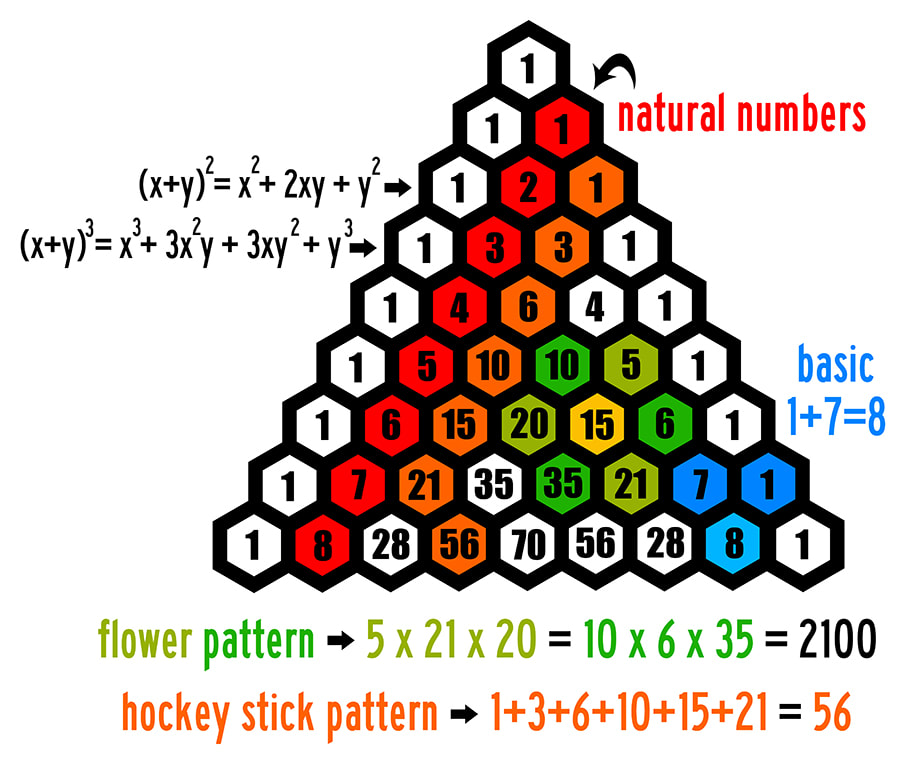
Physic love maths.
Pythagoras, born in 586 BC. in Samos, reaches Croton almost in 540 BC. and establishes the Pythagorean Battalion.
The Pythagoreans were interested, among other things, for the triangular numbers, the numbers that are today
are given by the formula:
Pythagoras, born in 586 BC. in Samos, reaches Croton almost in 540 BC. and establishes the Pythagorean Battalion.
The Pythagoreans were interested, among other things, for the triangular numbers, the numbers that are today
are given by the formula:
The sequence of triangular numbers is:
1, 3, 6, 10, 15, 21, 28, 36, 45, 55, 66, 78, ..........
The sequence of triangular numbers is found in each triangular seat of a normal tetrahedron. The set of balls of each seat is a sequence number.
1, 3, 6, 10, 15, 21, 28, 36, 45, 55, 66, 78, ..........
The sequence of triangular numbers is found in each triangular seat of a normal tetrahedron. The set of balls of each seat is a sequence number.
|
|
|
They are known to all the properties of the Pascal Triangle.
Watch the video below with the properties of the Pascal Triangle.
|
|
|
The first three sequences of numbers are expressed in the tetrahedron.
The sequence of natural numbers expresses the set of spheres in linear order.
The sequence of natural numbers expresses the set of spheres in linear order.
The sequence of triangular numbers (1,3,6,10,15,21,....), expresses the set of spheres in a flat (2D) configuration.
See the previous slideshow.
See the previous slideshow.
The sequence of tetrahedral numbers (1,4,10,20,35,56,.....) expresses the set of spheres in three-dimensional (3D) arrangement.
|
|
|
A GOOD QUESTION:
If the first three numbers sequences express the length, surface and volume, what would the next sequence of numbers represent ????????
If the first three numbers sequences express the length, surface and volume, what would the next sequence of numbers represent ????????
Download the Pascal triangle for the first 100x100 numbers.
| pascal_triangle.xlsx | |
| File Size: | 208 kb |
| File Type: | xlsx |
|
|
|
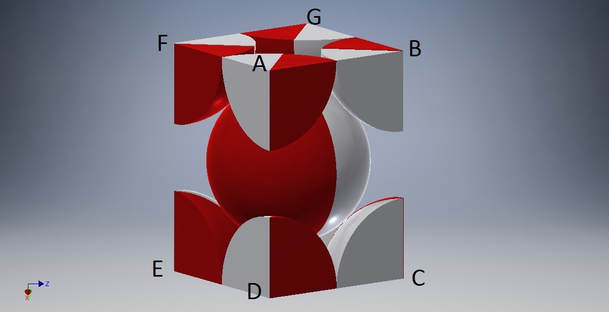
HEXAHEDRON
CUBE IN CCP
In this Hexahedron we observe that all sides have a simple packing arrangement of magnets.
However, the actual layout is at diagonal levels of the CCP type.
If we observe a square side we will see that each sphere is tangled with four spheres in the lower plane, four spheres in the same plane, and another four spheres in the upper plane, a total of 12 spheres. Thus, the cordination number is 12.
The unit cell expressing this type of crystallization is the Tetragonal (I) Body-Centered unit cell.
The crystallisation structure is CCP and the APF=0,74.
In this Hexahedron we observe that all sides have a simple packing arrangement of magnets.
However, the actual layout is at diagonal levels of the CCP type.
If we observe a square side we will see that each sphere is tangled with four spheres in the lower plane, four spheres in the same plane, and another four spheres in the upper plane, a total of 12 spheres. Thus, the cordination number is 12.
The unit cell expressing this type of crystallization is the Tetragonal (I) Body-Centered unit cell.
The crystallisation structure is CCP and the APF=0,74.
|
|
|
The nine spheres involved in the formation of the Tetragonal (Ι) Body-Centered unit cell.
|
|
|
To be clearer, the unit cell is expressed by the centers of the 8 spheres and they form a rectangular parallelepiped.
In the center of this rectangular parallelepiped there is a sphere. In this parallelepiped there are a total of two whole spheres,
8 Χ 1/8 + 1 = 2. So after calculations we can say that the APF=0,740. It is the same with the APF of the FCC unit cell.
In the center of this rectangular parallelepiped there is a sphere. In this parallelepiped there are a total of two whole spheres,
8 Χ 1/8 + 1 = 2. So after calculations we can say that the APF=0,740. It is the same with the APF of the FCC unit cell.
|
|
|
Calculation of the APF index.
|
If R is the radius of a sphere then:
AB= 2R BC= 2,828R AC= 3,464 FC= 4R The unit cell volume is: VUC = AB X AF X BC = 2R X 2R X 2,828R = 11,312 R*3 In this parallelepiped there are a total of two whole spheres, 8 Χ 1/8 + 1 = 2. VSPH = 2 X 4/3 X π R*3 = 8,377 R*3 APF = VSPH / VUC = 8,377 / 11,312 = 0,740 It is the same with the APF of the FCC unit cell. |
THE FCC (FACE CENTERED CUBIC) UNIT CELL AND THE TETRAGONAL BODY CENTERED (I) UNIT CELL IS THE SAME.
If we observe with caution the above Hexahedron (Cube in CCP), we will notice that on its sides coexist both unit cells.
Also, both cells have the same Atomic Packing Factor APF= 0,740.
So the question is asked: In which crystalline system the above crystal belong ? In Cubic or Tetragonal system?
I think it's best to place it in the Tetragonal system, because the Tetragonal Body Centered (I) unit cell is simpler.
It is made of only 9 spheres while the FCC unit cell of 14 spheres.
An important observation is also that all the crystal shape of the Tetragonal system ( Tetrahedron, Hexahedron, Octahedron, Cuboctahedron ), can be derived from the same original crystal shape, from the OBLIQYE HEXAGONAL PRISME, after incision in different crystallographic planes.
If we observe with caution the above Hexahedron (Cube in CCP), we will notice that on its sides coexist both unit cells.
Also, both cells have the same Atomic Packing Factor APF= 0,740.
So the question is asked: In which crystalline system the above crystal belong ? In Cubic or Tetragonal system?
I think it's best to place it in the Tetragonal system, because the Tetragonal Body Centered (I) unit cell is simpler.
It is made of only 9 spheres while the FCC unit cell of 14 spheres.
An important observation is also that all the crystal shape of the Tetragonal system ( Tetrahedron, Hexahedron, Octahedron, Cuboctahedron ), can be derived from the same original crystal shape, from the OBLIQYE HEXAGONAL PRISME, after incision in different crystallographic planes.
In the same group of spheres both the unit cells coexist. (The color section)
OCTAHEDRON
It is a very compact structure consisting of equilateral triangular planes with a closed magnet arrangement.
Each triangular plane has a Closed Packing (CP) structure.
The cordination number is 12.
The unit cell expressing this type of crystallization is the Tetragonal (I) Body-Centered unit cell.
The crystallisation structure is CCP and the APF=0,74.
More information about this polyhedron can be found at the link below:
en.wikipedia.org/wiki/Octahedron
Each triangular plane has a Closed Packing (CP) structure.
The cordination number is 12.
The unit cell expressing this type of crystallization is the Tetragonal (I) Body-Centered unit cell.
The crystallisation structure is CCP and the APF=0,74.
More information about this polyhedron can be found at the link below:
en.wikipedia.org/wiki/Octahedron
|
A different way of calculating the octahedron APF index.
|
|
|
The octahedron in the adjacent image consists of 146 beads. If R is the radius of a sphere then: The side of the octahedron is equal : α = 6 Χ 2R = 12R. The volume of the octahedron is equal: V = 0,471 α*3 = 0,471(12R)*3 The volume of the 146 beads is : Vs = 146 X 4/3 X π R*3 Therefore: APF = VS/V =611,563 R*3/813,888 R*3 = 0,751 |
CUBOCTAHEDRON
This structure can result from a regular octahedron by removing beads from its peaks.
We notice that in the triangular sides we have a Closed Packing (CP) sructure, while on the square sides we have an Simple Packing (SP) structure.
If we observe a square side we will see that each sphere is tangled with four spheres in the lower plane, four spheres in the same plane, and another four spheres in the upper plane, a total of 12 spheres. Thus, the cordination number is 12.
The unit cell expressing this type of crystallization is the Tetragonal (I) Body-Centred unit cell.
The crystallisation structure is CCP and the APF=0,74.
We notice that in the triangular sides we have a Closed Packing (CP) sructure, while on the square sides we have an Simple Packing (SP) structure.
If we observe a square side we will see that each sphere is tangled with four spheres in the lower plane, four spheres in the same plane, and another four spheres in the upper plane, a total of 12 spheres. Thus, the cordination number is 12.
The unit cell expressing this type of crystallization is the Tetragonal (I) Body-Centred unit cell.
The crystallisation structure is CCP and the APF=0,74.
|
|
|
More information about this polyhedron can be found at the link below:
en.wikipedia.org/wiki/Cuboctahedron
en.wikipedia.org/wiki/Cuboctahedron
OBLIQUE HEXAGONAL PRISME.
If we observe a rectagular side we will see that each sphere is tangled with four spheres in the lower plane, four spheres in the same plane, and another four spheres in the upper plane, a total of 12 spheres. Thus, the cordination number is 12.
The unit cell expressing this type of crystallization is the Tetragonal (I) Body-Centered unit cell.
The crystallisation structure is CCP and the APF=0,74.
|
|
|
Gold (Au), platinum (Pt) and silver (Ag) crystallize in the structure CCP. Also, many widely used metals such as copper (Cu), nickel (Ni), aluminum (Al), and lead (Pb) crystallize in this structure and are described by the FCC cell.
|
|
|
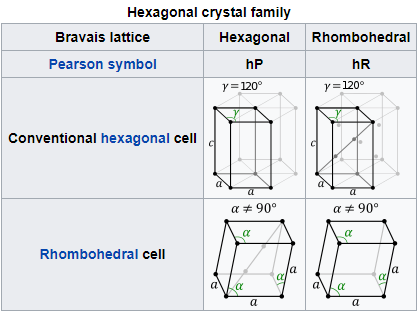
HEXAGONAL SYSTEM
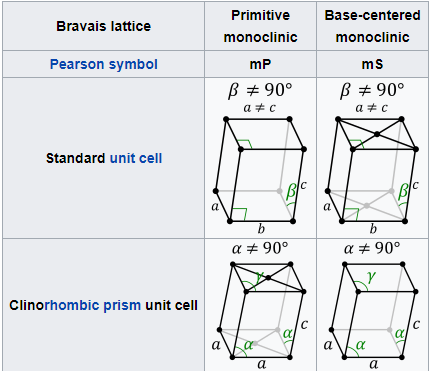
ORTHORHOMBIC SYSTEM
QUASICRYSTALS
Pentagonal bipyramid
It is a very compact structure consisting of equilateral triangular planes with a closed magnet arrangement.
Each triangular plane has a Closed Packing (CP) structure.
The crystallisation system is orthorhombic and the APF=0,7236
More information about this polyhedron can be found at the link below:
en.wikipedia.org/wiki/Pentagonal_bipyramid
Each triangular plane has a Closed Packing (CP) structure.
The crystallisation system is orthorhombic and the APF=0,7236
More information about this polyhedron can be found at the link below:
en.wikipedia.org/wiki/Pentagonal_bipyramid